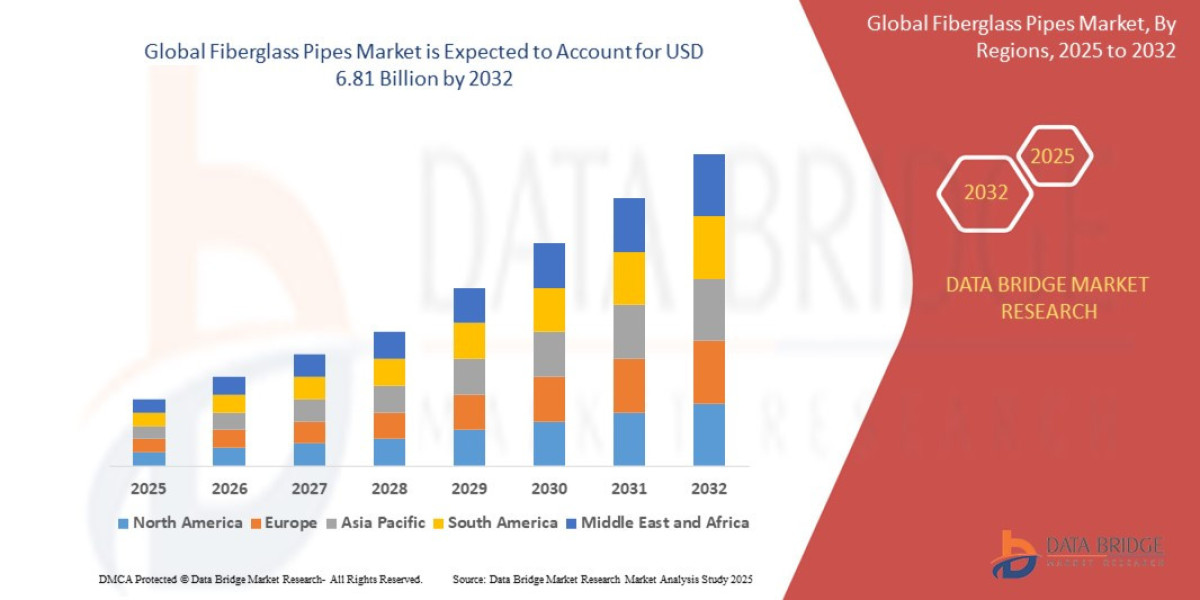
Pharmacovigilance outsourcing has become an integral component of modern drug safety systems, enabling pharmaceutical and biotechnology companies to efficiently monitor, assess, and report adverse drug reactions. As the global healthcare landscape evolves, the need for specialized expertise and advanced technologies in drug safety management is increasing significantly.
Expanding Drug Development Pipeline and Safety Requirements
The rapid growth in pharmaceutical research and development, including biologics and specialty drugs, has led to an increased volume of safety data that must be managed effectively. Clinical trials are becoming more complex and geographically diverse, necessitating robust pharmacovigilance systems. Outsourcing these functions helps companies manage large datasets while ensuring timely and accurate reporting. This trend is particularly beneficial for small and mid-sized firms that lack the internal infrastructure to handle comprehensive pharmacovigilance operations.
Access Comprehensive Market Insights: https://www.theinsightpartners.com/sample/TIPRE00040657
Technological Advancements Transforming Pharmacovigilance
Technological innovations such as artificial intelligence, machine learning, and automation are revolutionizing pharmacovigilance processes. These technologies enhance the efficiency of adverse event detection, improve signal analysis, and reduce manual errors. Outsourcing providers are leveraging cloud-based platforms and advanced analytics to deliver real-time insights and improve decision-making. This digital transformation is a key driver of growth, enabling pharmaceutical companies to adopt more efficient and scalable pharmacovigilance solutions.
Pharmacovigilance Outsourcing Market Trends and Growth Analysis
The Pharmacovigilance Outsourcing Market is experiencing strong growth, driven by increasing regulatory requirements, rising drug approvals, and the need for cost-effective safety solutions. According to insights from The Insight Partners, the market is expected to register a significant compound annual growth rate (CAGR) during the forecast period. North America dominates the market due to its advanced healthcare infrastructure and strong presence of pharmaceutical companies. Europe follows closely with strict regulatory frameworks, while the Asia-Pacific region is anticipated to witness the fastest growth due to cost advantages, a skilled workforce, and increasing outsourcing activities.
Regulatory Compliance and Global Safety Standards
Regulatory authorities across the globe are continuously strengthening pharmacovigilance guidelines to ensure patient safety. Compliance with these standards requires extensive documentation, monitoring, and reporting, which can be resource-intensive. Outsourcing pharmacovigilance functions allows companies to leverage the expertise of specialized providers who are well-versed in global regulatory requirements. This helps organizations reduce compliance risks and improve the efficiency of safety reporting processes.
Cost Efficiency and Operational Scalability
Cost optimization remains a major driver for pharmacovigilance outsourcing. Establishing and maintaining in-house pharmacovigilance capabilities involves significant investment in infrastructure, technology, and skilled personnel. Outsourcing provides a flexible and cost-effective alternative, allowing companies to scale their operations based on project requirements. This approach enhances operational efficiency and enables organizations to allocate resources more effectively.
Key Players in the Pharmacovigilance Outsourcing Market
Accenture plc
Cognizant Technology Solutions Corporation
IQVIA Inc.
Tata Consultancy Services Limited
Wipro Limited
Capgemini SE
Parexel International Corporation
Laboratory Corporation of America Holdings (LabCorp)
ICON plc
ArisGlobal
Pharmacovigilance Outsourcing Market Demand
The Pharmacovigilance Outsourcing Market Demand is increasing significantly due to the growing emphasis on drug safety and the rising complexity of regulatory requirements. Pharmaceutical companies are increasingly outsourcing pharmacovigilance functions to improve efficiency, reduce costs, and ensure compliance with global standards. The demand is further driven by the increasing adoption of real-world evidence and post-marketing surveillance, which require continuous monitoring and analysis. Additionally, the rise of personalized medicine and specialty drugs is creating a need for advanced pharmacovigilance solutions, further boosting demand across the industry.
Future Outlook and Emerging Opportunities
The future of the pharmacovigilance outsourcing market appears highly promising, with significant growth opportunities in emerging economies. Countries in Asia-Pacific and Latin America are becoming key outsourcing destinations due to their cost advantages and availability of skilled professionals. Companies are expected to invest in advanced technologies and expand their service portfolios to meet evolving industry needs. Strategic collaborations and partnerships will continue to shape the competitive landscape, driving innovation and enhancing service delivery.
Related Reports
About The Insight Partners
The Insight Partners delivers market intelligence and consulting services to help clients make informed decisions. The firm covers industries such as Aerospace and Defense, Automotive and Transportation, Semiconductor and Electronics, Biotechnology, Healthcare IT, Manufacturing, Medical Devices, Technology, Media, and Chemicals and Materials.
Contact Us
Email: sales@theinsightpartners.com
Website: www.theinsightpartners.com
Phone: +1-646-491-9876